 
Record physical state, color, luster and other observable characteristics.ģ. Observe the appearance of each of the elements. In your lab notebook, draw a table like the one shown below.Ģ. Write the whole property, not just the letter of the property.įor the observation stations you should prepare sealed test tubes containing the following elements:Ĭopper, silicon, magnesium, carbon, nickel, aluminum, zinc, sulfur, oxygen, lead, bismuth, silver, nitrogen, antimony, and hydrogenįor the conductivity station, you should have plastic dishes with one piece of each element to be tested and micro-conductivity testers.įor the malleability station, have one piece of each element per group along with paper towels and hammers.įor the reactivity station, have one piece of each element per group, 9 test tubes, test tube rack and 1 M HCl.ġ. Write each of the following properties under the appropriate heading. In you lab notebook, draw the following table. To investigate the properties of several elements on the periodic table and classify them as metals, non-metals or metalloids. A copy is also attached in the documents. This is a copy of the student handout of the lab. After everyone has completed the write-up, we will compare our results with the book classification and location of these elements. They will use this information to answer the analysis questions at the end of the lab. They will label each of these elements on a blank periodic table and color each group a different color. After they have completed their observations, they will determine if each element that they tested is a metal, non-metal or metalloid based on their observations. The stations can be completed in any order. Students will work in pairs or small groups and will rotate through the observation, conductivity, malleability and reactivity stations until they have completed all the stations. Students will be given the student handout (attached) and asked to write the experiment up in their lab notebooks. Before starting the lab, we will review the properties of each group. Although there is helium on Earth, it was first discovered by observing the sun.This lab activity will be used to have students discover the location of metals, non-metals and metalloids on the periodic table.The country Argentina is named after the element silver (symbol Ag) which is argentum in Latin.The only letter not in the periodic table is the letter J.There are probably no more than a few ounces of it on earth at any given time. Francium is the rarest element on earth.Carbon is important to the existence of life. Carbon is unique in that it is known to form up to 10 million different compounds.Using the table, Mendeleev was able to accurately predict the properties of many elements before they were actually discovered. The periodic table was proposed by Russian chemist Dmitri Mendeleev in 1869. For gold the "Au" comes from the Latin word for gold "aurum". Some are a bit harder like Fe for iron or Au for gold. Some of the abbreviations are easy to remember, like H for hydrogen. They can understand and predict how an element might react or behave in a certain situation.Įach element has its own name and abbreviation in the periodic table. This lining-up and grouping of similar elements helps chemists when working with elements. You can see all the groups in the table below. They are all very similar in that they have only 1 electron in their outer shell and are very reactive. Another example is the alkali metals which all align on the left-most column. They all have a full outer shell of electrons, making them very stable (they tend not to react with other elements). These elements all line up in the eighteenth or last column of the periodic table. One example of a group is the noble or inert gases. 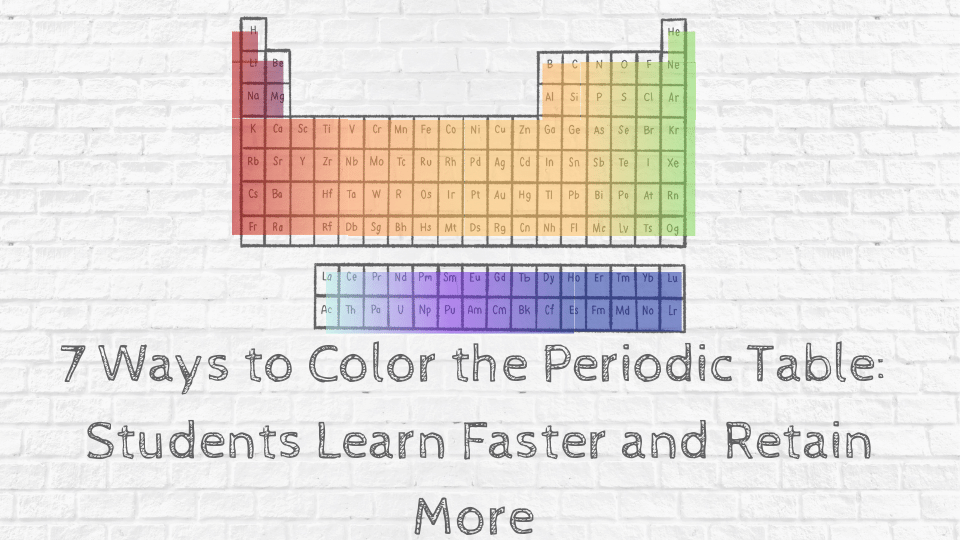
There are 18 columns or groups and different groups have different properties. 
Groups are the columns of the periodic table. 
In each period the left most element has 1 electron in its outer shell and the right most element has a full shell. The first one is short and only has two elements, hydrogen and helium. There are seven (or eight) total periods. When they are lined up this way, elements in the columns have similar properties.Įach horizontal row in the table is a period. Some columns are skipped in order for elements with the same number of valence electrons to line up on the same columns. From left to right elements are lined up in rows based on their atomic number (the number of protons in their nucleus). It is called "periodic" because elements are lined up in cycles or periods. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
